LABM
An antagonist-free medium for use in the tube dilution technique for the determination of antibiotic M.I.C. values.
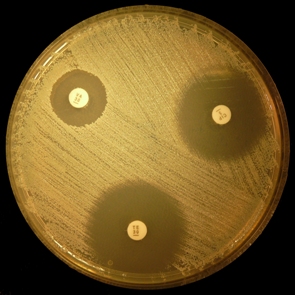
A medium for antimicrobial sensitivity testing by the disc diffusion method. This medium, used in the technique of Bauer and Kirby, has been adopted by the National Committee for Clinical Laboratory Standards (NCCLS) in the USA as the definitive method for susceptibility testing. The medium has a very low thymine and thymidine content, making it suitable for trimethoprim and sulphonamide testing, controlled to ensure correct zone sizes with aminoglycoside and tetracyline antibiotics. The medium was originally formulated as a heat labile protein free medium for the isolation of pathogenic Neisseriaceae.
A semi-solid medium, based on Rappaport Vassiliadis broth, for the rapid detection of motile Salmonella, originally developed in 1986 by De Smedt, Bolderdijk and Rappold.
A semi-solid medium, based on Rappaport Vassiliadis broth, for the rapid detection of motile Salmonella, originally developed in 1986 by De Smedt, Bolderdijk and Rappold.
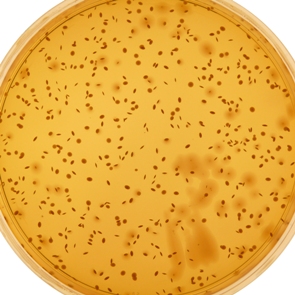
MRS Agar (ISO) is a medium for the enumeration of mesophilic lactic acid bacteria according to ISO 15214:1998.
This medium was originally developed in 1960 by de Man, Rogosa & Sharpe for the cultivation and enumeration of Lactobacillus spp. from various sources and is intended as a substitute for Tomato Juice Agar. This original formulation has been adapted and adjusted to pH 5.7 according to ISO 15214:1998.
Nutrition is provided by enzymatic digest of casein, glucose, meat & yeast extracts whilst polyoxyethylenesorbitan monooleate, magnesium and manganese sulphates act as growth stimulants. Selectivity against streptococci & moulds is provided by ammonium citrate and sodium acetate. Used at low pH, ammonium citrate allows growth of lactobacilli whilst inhibiting a number of other organism groups.
Although MRS Agar (ISO) is optimised to be selective for Lactobacilli, some growth of Leuconostoc spp. and Pediococci may also occur.


.jpg)